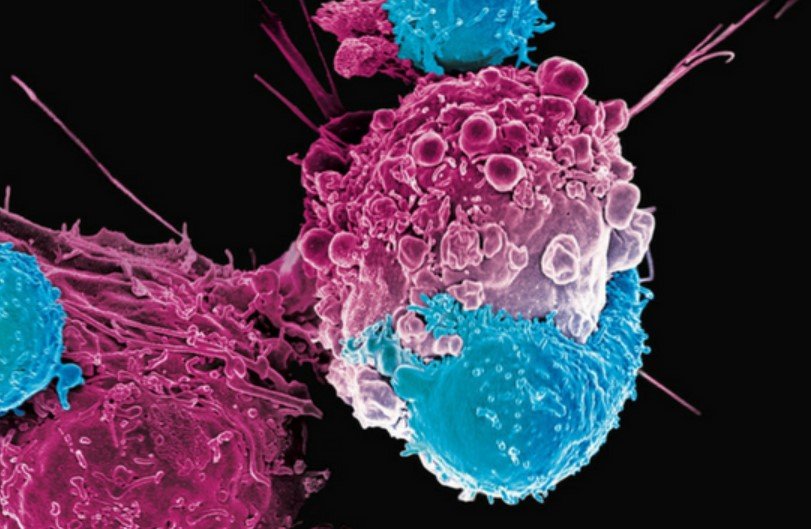
Beatty Laboratory
The Beatty Laboratory incorporates both basic science research and clinical investigation to define strategies for leveraging the immune system for the treatment of cancer. Our mission is to define mechanisms of resistance and response to immunotherapy in an effort to design novel effective strategies that can improve outcomes for patients. Support our program.
Pancreatic Cancer
In 2021, an estimated 60,430 people are predicted to be diagnosed with pancreatic cancer in the United States and 48,220 are predicted to die from their disease. Diagnoses and deaths continue rise yearly. The 5-year survival remains at 10%. Together, these alarming statistics emphasize the continued need for further advancements aimed at improving clinical outcomes.
Immunotherapy
The lab’s main focus is pancreatic cancer, a malignancy that has demonstrated remarkable resistance to all forms of therapy, including immunotherapy. A key determinant of this resistance is the microenvironment that surrounds cancer cells. We believe that understanding this biology will inform novel strategies to “condition” "cancer for successful “treatment” and that this knowledge will have implications not only for pancreatic cancer but also many other malignancies.
Machine Learning
A major goal within the laboratory is to understand the “connectome” of immune cells within tumors and to identify mechanisms that define the biology of these immune cell communities. We are using sophisticated models to discover novel treatment algorithms, collaborating within clinical investigators to understand human biology within metastatic and primary tumors, and working to revolutionize and advance novel therapeutics into the clinic.